
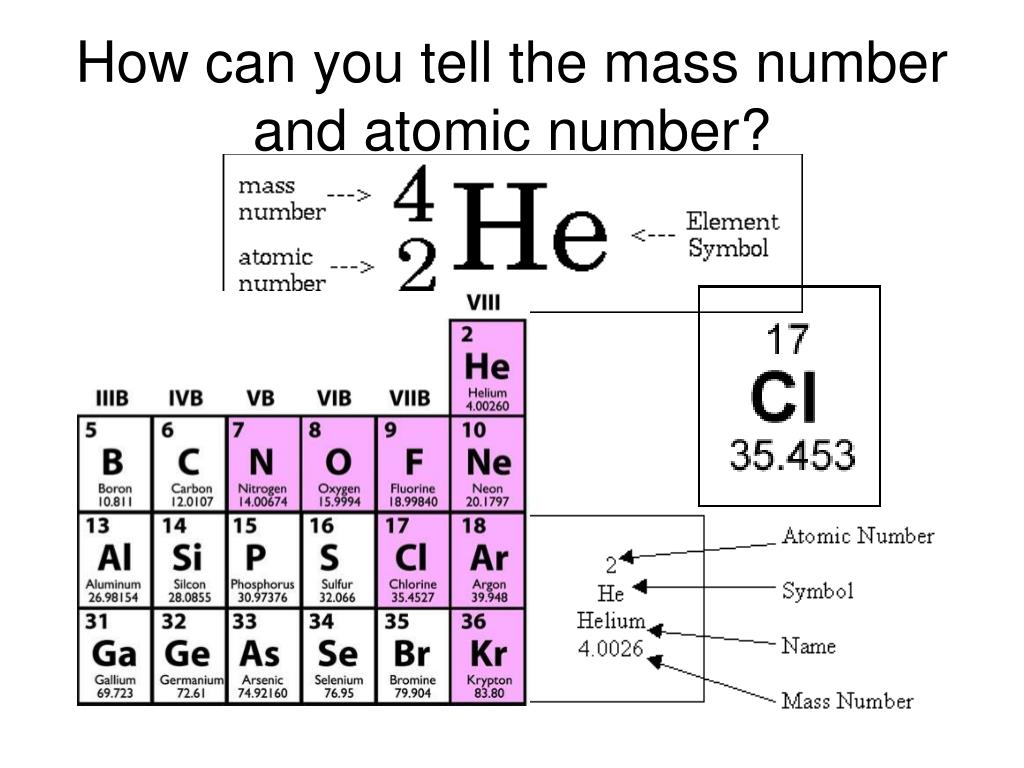
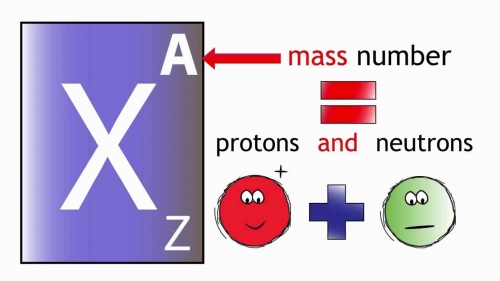
Atomic Number and the Number of Neutrons. We know that mass number is the sum of the number of protons i.e. The formula used for calculating Mass Number can be modified to calculate the number of protons. Atomic Number and the number of neutrons to get the Mass Number of the element. Step 2: Now look for the number of neutrons in the atom. Step 1: First look for the Atomic Number of the element as the Atomic Number is equal to the mass number. Mass Number can be calculated using the following steps: Mass Number of an atom is the total number of protons and neutrons present in the mass number. Examples include Ar-40 and Ca-40 both have the same mass number but different atomic numbers as they are different elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
:max_bytes(150000):strip_icc()/atomic-mass-and-mass-number-606105_v1-80df956ab98440bc9969531d1bb6c874.png)
 RSS Feed
RSS Feed
